Ethics and governance
Metro North Hospital and Health Service (MNHHS) is committed to the highest standards of research integrity. Research governance, including the ethical review of research, refers to the processes to ensure that research in Metro North Hospital and Health Service is conducted according to the appropriate regulatory, ethical and scientific standards. The framework for research governance is relevant to all stages of the research process.
Research approvals
In order to conduct research in MNHHS, it is a requirement that all research first obtains the following:
- Ethics approval from a Human Research Ethics Committee (HREC)
- Site authorisation at each facility you are intending to do the research.
Ethics approval is required before Site Authorisation can be granted. However, MNHHS, encourages parallel submission for research ethics and governance applications.
The MNHHS research ethics committees and research governance officers are the contact point for obtaining relevant research approvals. Research Ethics and Research Governance application forms are prepared and submitted through ERM.
Use of confidential health information
In some cases approval to use confidential information for the purposes of research must be obtained after ethics approval and prior to site authorisation.
Post-approval reporting
While undertaking a research project, researchers have an obligation to both the participants and to the Human Research Ethics Committee (HREC) and Research Governance Officers to provide reporting and monitoring.
Metro North Research policy and procedures
The Metro North Research Policy and Procedures provide a framework to promote the responsible and ethical design, conduct and communication of research. Each is based on the principles of the National Statement on Ethical Conduct in Human Research (National Statement) and the Australian Code for the Responsible Conduct of Research (the Code), in the context of institutional policies, state and federal legislation and regulatory guidelines.
Frequently asked questions
General
The Australian Code for the Responsible Conduct of Research defines research as “original investigation undertaken to gain knowledge, understanding and insight”. In terms of illustrating what the term ‘research’ can cover, the Code and the National Statement on Ethical Conduct in Human Research quote the following definition for research:
‘Research’… includes work of direct relevance to the needs of commerce, industry, and to the public and voluntary sectors; scholarship; the invention and generation of ideas, images, performances, artefacts including design, where these lead to new or substantially improved insights; and the use of existing knowledge in experimental development to produce new or substantially improved materials, devices, products and processes, including design and construction. It excludes routine testing and routine analysis of materials, components and processes such as for the maintenance of national standards, as distinct from the development of new analytical techniques. It also excludes the development of teaching materials that do not embody original research.
The National Statement defines human research as being “conducted with or about people, or their data or tissue”. Human participation in research is therefore to be understood broadly, to include the involvement of human beings through:
- taking part in surveys, interviews or focus groups;
- undergoing psychological, physiological or medical testing or treatment;
- being observed by researchers;
- researchers having access to their personal documents or other materials;
- the collection and use of their body organs, tissues or fluids (eg: skin, blood, urine, saliva, hair, bones, tumour and other biopsy specimens) or their exhaled breath;
- access to their information (in individually identifiable, re-identifiable or non-identifiable form) as part of an existing published or unpublished source or database.
The benchmark for receipt of an ethical decision is 60 days from receipt of a valid ethics application. The benchmark for site authorisation is 25 days from receipt of a valid Site Specific Assessment form.
Low or negligible risk research studies are typically reviewed out of session of the HREC with a response received within 4 weeks of submission. For studies requiring HREC review, you should expect to receive a response within 2 weeks of the HREC meeting date. Site authorisation is typically received within 2 weeks of submission of a valid Site Specific Assessment form.
To reduce delays associated with incomplete Site Specific Assessment submissions, researchers are encouraged to make early contact with the relevant Research Governance Officer and undertake completion of the Site Specific Assessment form in parallel with ethical review.
Fees
Applicable Ethics and Governance Fees until 30 April 2024.
| Fee type | Fee schedule (inclusive of GST) |
|---|---|
| HREC fees for application for research project with full industry sponsorship | $4500 |
| HREC fees for major amendments to research projects with full industry sponsorships | $650 |
| HREC fees for addition of sub-studies to research projects with full industry sponsorship | $2000 |
| Site-Specific Assessment (SSA) Fee | $4500 per site |
| Research Governance fee for major amendments to research projects with full industry sponsorships | $650 |
If you have any queries regarding fees, please contact the HREC Coordinator or Research Governance Officer.
Applicable Ethics and Governance Fees from 1 May 2024.
From May 1, the following Schedule of Fees applies.
The completed and signed fee template must be provided with the relevant ethics/research governance submission via Ethical Review Manager (ERM). Failure to provide the form at time of submission will delay review and may result in the submission being withdrawn from review.
- MNH Research Fee Template – This fee template applies for all submissions that are not sponsored or led by a Queensland Health Hospital and Health Service (HHS).
- MNH Research Fee Template HHS – Inter HHS Journal Transfer (applies only for other Queensland Health Hospital and Health Services).
New fees will only apply to new submissions post 1 May 2024, with the exception of commercially sponsored projects, whom the updated fee structure will apply to new and existing submissions.
Yes. Fees apply to the institution that is leading the research collaborative project.
Fees are applied to the institution leading the research project. If a university is leading the project, then yes, fees will apply.
Yes. Fees apply as you are conducting the research as a university student, not a Metro North Health employee. If a Metro North Health employee is undertaking a research higher degree with a university, this is classified as a investigated-initiated externally led study and is subject to a fee. This is because the research higher degree student is representing the University.
This is research that is initiated by a non-pharmaceutical company and led by the Chief/Coordinating Principal Investigators institution such as a university or hospital. Fees apply for all investigator-initiated studies where the Coordinating Principal Investigator (CPI) is from an institution external to Metro North Health.
A Metro North Health investigator-initiated/led study is one where the CPI is representing Metro North Health. Fees do not apply if the study is led/sponsored by Metro North Health.
Fees apply for research that is not led by Metro North Health. Your application to the HREC or research governance should clearly detail which organisation is leading/sponsoring the research project.
No, fees will only apply for new CRG sponsored studies (and subsequent amendments) submitted post 1 May 2024.
No, fees will only apply for new investigator-initiated sponsored studies (and subsequent amendments) submitted post 1 May 2024.
Yes, fees apply for post approvals depending upon the research and submission type. Please refer to the respective schedule of fees.
Major amendments include the following:
- Changes to the protocol and/or Participant Information and Consent Form (PICF) beyond administrative changes
- Changes that require review by the ethics committee, sub-committee or other expert/legal review
- Review of a sub-study
- Addition of a site
- Changes to investigators that require study document and/or agreement/indemnity updates.
- Other agreement/contract amendments/variations
- Changes to procedures undertaken by participants.
- Updates to Investigator’s Brochure.
- Administrative changes e.g., typographical error correction, version control
- Minor changes to the protocol or other study documentation e.g. correcting errors, minor clarification
No, if fees apply to your submission, the completed and signed fee template must be provided. Failure to provide the form at time of submission will delay review and may result in your submission being withdrawn from review.
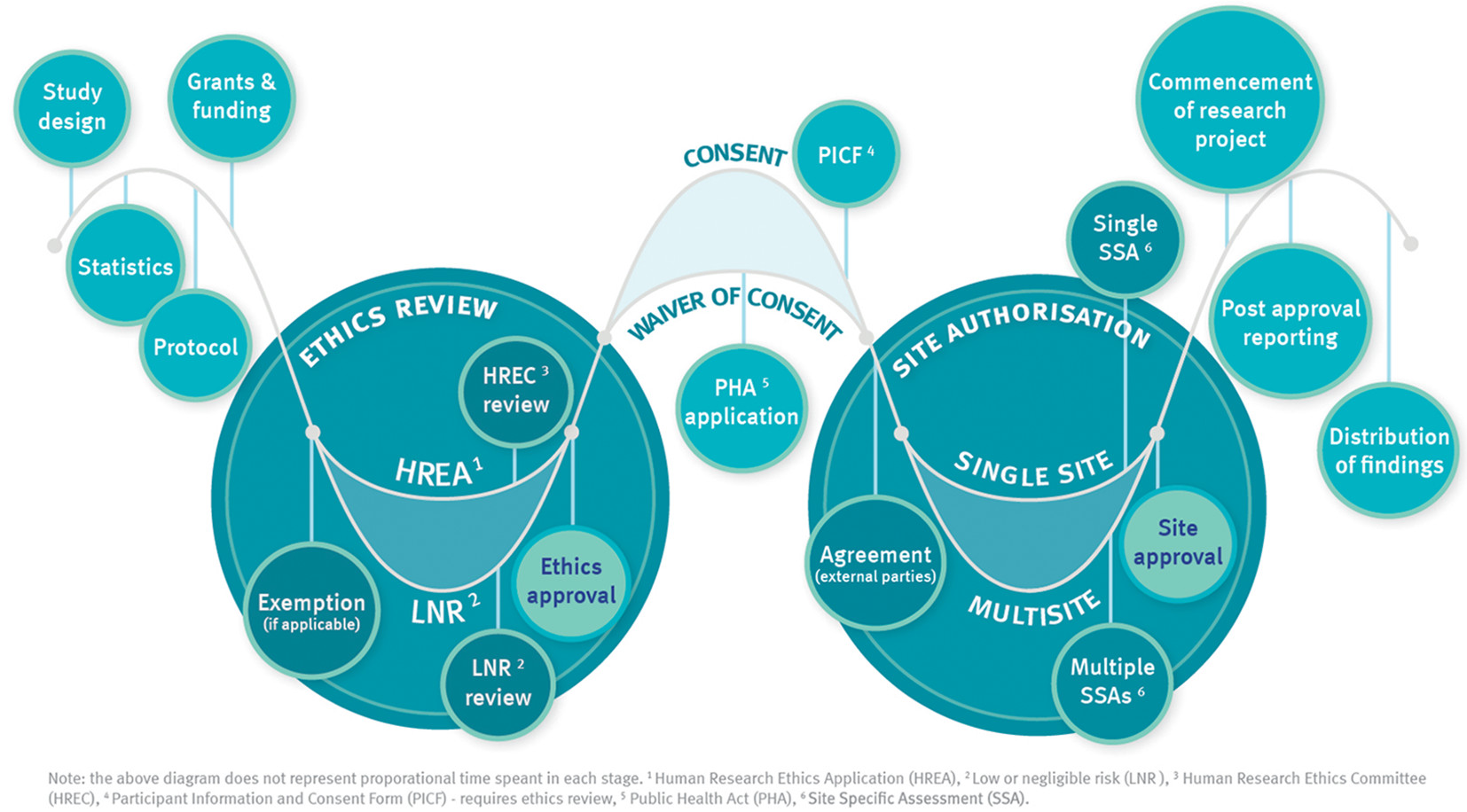
Research process
Key steps in the research approval process are shown in the diagram below.

RIPE program embeds strong research culture
The Research in Practice (RIPE) program was developed to address a gap in research skills and experience among the prevocational doctor workforce at Caboolture Hospital.


