Dengue
Queensland Health Guidelines for Public Health Units
Revision History
| Version | Date | Changes |
| 1.0 | January 2011 | Full revision of guideline |
| 2.0 | January 2017 | Case definition revision |
Infectious Agent
Dengue virus (DENV) is a member of the Flavivirus genus; it is classified into four serotypes, dengue 1 to dengue 4.
Each of the four serotypes is capable of causing the full spectrum of clinical manifestations of dengue. Infection with one serotype leads to life-long immunity to that particular serotype.
Notification Criteria
Clinical evidence
A clinically compatible illness.
Laboratory suggestive evidence1
Detection of dengue IgM in serum by screening ELISA test. This antibody may cross react with other flaviviruses or be falsely positive. It requires confirmation with further tests at a reference laboratory. False negative IgM occurs in early illness - see Testing Guidelines below.
Confirmed case
A confirmed case requires laboratory definitive evidence AND clinical evidence
Laboratory definitive evidence
Isolation of dengue virus
OR
- Detection of dengue virus by nucleic acid testing
OR
- Detection of non-structural protein 1 (NS1) antigen in blood by EIA
OR
- IgG seroconversion or a significant increase in antibody level or a fourfold or greater rise in titre to dengue virus, proven by neutralisation or another specific test
OR
- Detection of dengue virus-specific IgM in cerebrospinal fluid, in the absence of IgM to Murray Valley encephalitis, West Nile virus /Kunjin, or Japanese encephalitis viruses
Clinical evidence
A clinically compatible illness (e.g. fever, headache, arthralgia, myalgia, rash, nausea/vomiting)
Probable case
A probable case requires:
- Laboratory suggestive evidence and clinical evidence and epidemiological evidence
OR
- Clinical evidence and household epidemiological evidence
Laboratory suggestive evidence
- Detection of NS1 antigen in blood by a rapid antigen test2
OR
- Detection of dengue virus-specific IgM in blood
Epidemiological evidence
- Exposure, between 3 and 14 days prior to onset, in
Either
- a country with known dengue activity
OR
- a dengue-receptive area3 in Australia where a locally-acquired or imported case has been documented with onset within a month
Household epidemiological evidence
- Living in the same house4 as a locally-acquired case in a dengue-receptive area3 of Australia within a month of the onset of the case.
AND
- At least one case in the chain of epidemiologically linked laboratory-confirmed cases (which may involve many cases).
- Confirmation of the laboratory result by an arbovirus reference laboratory is required if the infection was acquired in Australia but outside a dengue-receptive area as defined in the Dengue National Guideline for Public Health Units.
- Unless dengue NS1 antigen by EIA is negative.
- As defined in the Dengue CDNA National Guideline for Public Health Units.
- The case must have spent all the exposure period (from 14 days prior to onset to 3 days prior to onset) living in the same house as the epi-linked confirmed case.
Notification Procedure
Pathology Laboratories
Notify on laboratory definitive or suggestive evidence, immediately by telephone or facsimile if the case is in north Queensland or where Aedes aegypti is known to be present, or by usual means in other areas.
Attending Medical Practitioners/Medical Superintendents (or delegates)
Notify clinically suspected cases to the relevant local public health unit immediately by telephone or facsimile, before laboratory results become available, if the case is in north Queensland, or where Aedes aegypti is known to be present.
Reporting to NOCS
Both confirmed cases and probable cases should be notified.
Objectives of surveillance
- Early detection and response to locally acquired cases of dengue in Queensland.
- Early detection and response to viraemic importations of DENV into north Queensland and other receptive areas.
- To monitor the epidemiology of dengue and its vector mosquitoes in Queensland.
Public Health Significance and Occurrence
Dengue is an acute febrile viral disease that occurs throughout much of the tropics. It is the most rapidly spreading mosquito-borne viral disease globally; Southeast Asian and South Pacific island nations have been severely affected in recent years. Dengue has a wide clinical spectrum; severe complications can be fatal (shock from plasma leakage, severe haemorrhage or organ failure).
Although not endemic in Australia, DENV is frequently imported from abroad. Because the main mosquito vector of DENV, Aedes aegypti, is established in north Queensland coastal towns, this region is particularly receptive to local transmission following viraemic importations of DENV. Outbreaks of dengue in north Queensland have become more frequent since the early 1990s; some have been very large and have placed considerable demands on clinical, diagnostic, disease control and vector control services.
Aedes aegypti has also been detected in towns as far south as Goomeri near the coast and Charleville inland. Historically it was also present in other parts of Queensland and in other Australian states and territories. Large outbreaks were documented in Brisbane up to the 1940s. Changes in vector range and numbers could increase the area receptive to DENV outbreaks. This depends on both climate and water holding practices - for example, increased reliance on home water tanks in drought prone towns.
Ae. albopictus, an exotic and less efficient vector, has colonised the majority of islands in the Torres Strait since 2005, and on some islands appears to have displaced Ae. aegypti. A small incursion on the northernmost tip of Cape York as of January 2010, highlights the risk of dengue transmission expanding within Queensland and interstate, as this mosquito species could potentially colonise areas as far south as Melbourne (see below).
Clinical Features
The World Health Organization recently (2009) revised the dengue case classification into two main categories:
- Dengue with or without warning signs
Probable dengue is defined by residence in or travel to a dengue endemic (or epidemic) area, fever and two of the following:- nausea, vomiting
- rash
- headache
- arthralgia, myalgia
- a positive tourniquet test (indicative of low platelet count)
- low leycocyte count
- abdominal pain or tenderness
- persistent vomiting
- clinical fluid accumulation
- mucosal bleeding
- lethargy, restlessness
- liver enlargement >2 cm
- an increase in the haematocrit concurrent with a rapid fall in the platelet count.
- Severe dengue
Any of the following:- Severe plasma leakage leading to:
- shock (dengue shock syndrome); or
- fluid accumulation with respiratory distress
- severe bleeding (as evaluated by the attending clinician)
- severe organ involvement affecting:
- liver (AST or ALT ≥1000; or
- CNS (impaired consciousness); or
- heart; or
- other organs.
- Severe plasma leakage leading to:
Reservoir
Human and Aedes mosquitoes (sylvatic cycles, involving monkeys and Aedes mosquitoes, occur in Africa).
Mode of Transmission
Bites from infected females of certain Aedes mosquito species. Ae. aegypti is the major vector of dengue; Aedes albopictus can also transmit DENV, but much less efficiently than Ae. aegypti.
Ae. aegypti is a very domesticated, urban mosquito found in the tropics and subtropics, and in Australia is currently confined to Queensland; it usually breeds in man-made containers and prefers indoor, sheltered, dark resting sites. Humans are the preferred source of blood meals for female Ae. aegypti; they are day-biting mosquitoes.
Ae. albopictus is a peri-domestic mosquito found not only in the tropics and subtropics but also some temperate regions. It is an aggressive coloniser, but in Australia is currently confined to the Torres Strait. It breeds not only in artificial containers but also in some naturally occurring sites such as tree holes and coconut shells. Adults prefer heavily-shaded outdoor resting sites; the female takes blood from a wide range of mammals. It is an aggressive day-biting mosquito.
Incubation Period
Following the bite of an infected mosquito, the incubation period in a susceptible human (ie. the intrinsic incubation period) is usually 4-7 days, but may range from 3-14 days.
After feeding on a viraemic human, DENV replicates within the female mosquito and then spreads systemically. The mosquito becomes infectious 8-12 days after ingesting DENV. This extrinsic incubation period is influenced by environmental conditions, especially the ambient temperature.
The sum of the extrinsic and intrinsic incubation periods indicates a minimum serial interval of approximately two weeks between generations of human cases
Period of Communicability
A human case is infective to mosquitoes from shortly before, until the end of the period of symptomatic viraemia; usually 4 - 5 days, but can be longer.
For public health purposes, the duration of viraemia is assumed to be from one day before until 12 days after the onset of symptoms.
There is no direct person to person transmission, but transfusion related cases can occur.
Susceptibility and Resistance
Susceptibility to primary infection appears universal. Recovery from infection with one DENV serotype provides lifelong immunity against that serotype, but only short-term protection against other serotypes.
Antibody dependent enhancement (ADE) is an immunological phenomenon believed to be responsible for the pathogenesis of plasma leakage, a potentially severe manifestation of dengue. In brief, following infection with a particular DENV serotype, the cross-protection against other serotypes wanes after several months; at some point the previously protected person has, through ADE, an increased risk of developing severe dengue following a secondary infection (from another DENV serotype).
It is hypothesised that ADE occurs in the secondary infection when pre-existing, but no longer protective, antibodies enhance the binding of the (secondary) virus to macrophages and monocytes, leading to increased virus replication in these cells. This, in turn, leads to complement activation and the release of various cytokines that target vascular endothelial cells, resulting in the leak of plasma and protein.
Testing Guidelines
Serology
- Dengue IgM testing is of limited use very early in the illness, as it only becomes detectable between about day 3 and day 7 (see Figure). IgM detected by screening ELISA or rapid antibody test is often a false positive or cross–reaction. IgM needs to be confirmed by more specific testing at a reference laboratory.
- In secondary dengue (reinfection with a different serotype), IgG rises steeply a few days after onset, often with minimal or transient IgM. A rise in IgG (often reflecting the original rather than reinfection serotype) should be confirmed by a reference laboratory.
- After the viraemic period, serology is not always definitive on a single specimen. A second sample may be required, 10-14 days after the first, to detect seroconversion or exclude cross-reaction with another flavivirus.
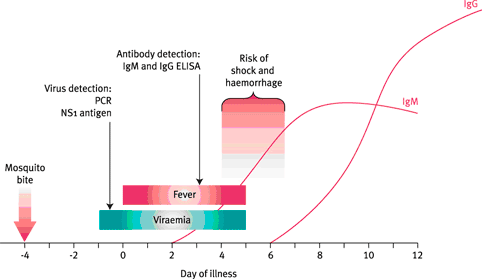 |
| Figure: Typical primary dengue infection with timing of diagnostic tests |
PCR
- Nucleic acid testing detects dengue early in the illness (see Figure) during the viraemic phase, which usually lasts to day 5 but sometimes longer, occasionally to day 9.
Viral antigen
- Non-structural protein 1 (NS1) is also detectable early in the illness (see Figure).
NS1 and/or PCR testing is vital in early cases of suspected dengue, because antibody may not be present. Early diagnosis is important for rapid dengue control measures.
Management
Cases
Investigation
Determine the likely location(s) of exposure to DENV (i.e. where the infection was presumably acquired), and addresses where the case lived/worked/visited while viraemic (for public health purposes this is assumed to be from one day before until 12 days after the onset of symptoms).
Ensure specimens are taken or referred for confirmation of the case. Determine on which day of illness the sample was taken, and ensure the appropriate tests are performed. In practice, if in the first week of illness this means dengue PCR and NS1 tests should be included.
Management
If a case considered viraemic stays in, or travels to, an area where Aedes aegypti are present, particularly in north Queensland:
- Share appropriate case information between local communicable disease, environmental health and/or medical entomology senior staff.
- Advise the case and household contacts to prevent bites and kill mosquitoes for at least as long as the case is febrile and unwell. Persons geographically close to the case who develop symptoms should see their doctor for possible dengue testing.
- Consider urgent house to house mosquito control and surveys to detect further cases, in conjunction with local government.
- Consider alerts to general practitioners, laboratories, emergency departments and the public.
Contacts
Contact Tracing
Yes
Ask patients if relevant co-travellers, family members, work colleagues, etc are sick. Do follow up interviews with these and arrange testing if dengue is suspected.
Community outbreaks/epidemics
In an outbreak, an incident management team including a medical entomologist should be formed. Significant disease surveillance, mosquito control and educational measures will be required. Refer to current dengue management plans.
Blood donation services in outbreak locations may be restricted, so liaison with the Australian Red Cross Blood Service is important.
Preventive Measures
Travellers to tropical countries should take precautions to prevent dengue:
- ensuring accommodation is free of mosquitoes, by closing window screens, using insecticide sprays indoors, etc.
- wearing long sleeved clothing in urban or residential areas to minimise skin exposure to day-biting mosquitoes
- using an appropriate mosquito repellent on exposed skin.
Someone who becomes unwell with a high fever during, or soon after, travel to tropical destinations should seek medical advice as soon as is practicable. If the person is told that they may have dengue, the person should use a mosquito repellent and stay in well screened premises for at least as long as the fever persists.
If the person is in coastal north Queensland, or other areas where Aedes aegypti are present, mosquito control and other preventive measures may be required.
For further information on preventive measures, refer to current dengue fever management plans.
Summary
Prepare a report of the investigation for the Communicable Diseases Branch, Queensland Health on request.
References
Queensland Dengue Management Plan 2015>2020. Queensland Health, Communicable Diseases Branch. 2015: https://www.health.qld.gov.au/__data/assets/pdf_file/0022/444433/dengue-mgt-plan.pdf
Heymann D (Ed). 2015. Control of Communicable Diseases Manual, 20th edition. American Public Health Association: Washington.
World Health Organization and the Special Programme for Research and Training in Tropical Diseases. Dengue: guidelines for diagnosis, treatment, prevention and Control. Geneva; World Health Organization: 2009:
http://www.who.int/tdr/publications/documents/dengue-diagnosis.pdf?ua=1