Rubella
Queensland Health Guidelines for Public Health Units
Disclaimer
Infectious Agent
Case Definitions and Notification Criteria - Rubella
Case Definitions and Notification Criteria - Congenital Rubella Syndrome
Notification Procedure
Objectives of Surveillance
Public Health Significance and Occurrence
Clinical Features
Reservoir
Mode of Transmission
Incubation Period
Period of Communicability
Management
Preventive Measures
References
Appendices
Revision History
Infectious Agent
Rubella virus is a togavirus of the genus Rubivirus.
Case definitions and notification criteria
Rubella can present as Rubella infection, congenital rubella infection or congenital rubella syndrome.
Rubella
Report confirmed and probable cases.1
Confirmed case
A confirmed case requires laboratory definitive evidence only.
Laboratory definitive evidence
- Isolation of rubella virus*
OR
- Detection of rubella virus by nucleic acid testing*
OR
- IgG seroconversion or a significant increase in antibody level, such as a fourfold or greater rise in titre to rubella virus EXCEPT if the case has received a rubella-containing vaccine eight days to eight weeks prior to convalescent specimen collection. (NOTE: paired sera must be tested in parallel).
*Where rubella vaccine has been given in the 3 weeks prior to illness onset and wild-type virus is not detected, or unable to be detected, a case may be considered “Probable” only if the criteria for clinical and epidemiological evidence can also be met, suggesting wild-type infection. Vaccine-associated rubella illness (genotype 1A) is not notifiable, but rather should be reported as an adverse event following immunisation.
Probable case
A probable case requires:
- Laboratory suggestive evidence AND clinical evidence
OR
- clinical evidence AND epidemiological evidence.
Laboratory suggestive evidence
Detection of rubella-specific IgM antibody**, EXCEPT
- if ruled out by more specific rubella IgM serology testing at a jurisdictional public health laboratory#
OR
- if the case has received a rubella-containing vaccine 8 days to 8 weeks before testing.
Clinical Evidence
- A generalised maculopapular rash
AND
- Fever
AND
- Arthralgia/arthritis OR lymphadenopathy OR conjunctivitis.
Epidemiological evidence
An epidemiological link is established when there is:
Contact between 2 people involving a plausible mode of transmission at a time when:
- one of them is likely to be infectious (about one week before to at least 4 days after appearance of rash)
AND
- the other has an illness that starts within 14 and 23 days after this contact
AND
- at least one case in the chain of epidemiologically linked cases (which may involve many cases) is laboratory confirmed.
**see Laboratory Aspects section.
#not available at Queensland Health Forensic and Scientific Services laboratory.
Congenital Rubella Infection
Congenital rubella infection is reported based on relevant evidence from a live or stillborn infant, miscarriage or pregnancy termination. Congenital rubella syndrome is reported as a subset of congenital rubella infection.
Both confirmed cases and probable cases should be notified.2
Confirmed case
- A confirmed case requires laboratory definitive evidence (fetal).
OR
- laboratory definitive evidence (infant) AND epidemiological evidence.
Laboratory definitive evidence
Fetal
Isolation or detection of rubella virus from an appropriate clinical sample (i.e. fetal blood or tissue, amniotic fluid, chorionic villus sample) by culture or nucleic acid testing.
Infant
- Isolation or detection of rubella virus from an appropriate clinical sample in an infant, by culture or nucleic acid testing
OR
- detection of rubella-specific IgM antibody in the serum of the infant.
Epidemiological evidence
The mother has confirmed rubella infection during pregnancy (see definition for Rubella, non-congenital).
Probable case
A probable case requires:
- Epidemiological evidence (first trimester infection)
OR
- epidemiological evidence (second and third trimester infection) AND laboratory suggestive evidence (infant).
Laboratory suggestive evidence
Infant
High/rising rubella-specific IgG level in first year of life.
Congenital Rubella Syndrome
Both confirmed cases and probable cases should be reported.2
Confirmed case
A confirmed case requires laboratory definitive evidence (foetal or infant), as described above AND clinical evidence.
Clinical evidence
A live or stillborn infant with ANY of the following compatible defects:
- Cataracts
- Congenital glaucoma
- Congenital heart disease
- Hearing defects
- Microcephaly
- Pigmentary retinopathy
- Development delay
- Purpura
- Hepatosplenomegaly
- Meningoencephalitis
- Radiolucent bone disease
- Other defect not better explained by an alternative diagnosis.
Probable case
A probable case requires laboratory suggestive evidence (infant) OR epidemiological evidence, as described above AND clinical evidence (as for confirmed Congenital Rubella Syndrome case).
Notification Procedure
Pathology laboratories
To notify on microbiological or serological confirmation of rubella or congenital rubella syndrome, by usual means.
Laboratory aspects
IgM is produced soon after vaccination, reaching a peak at one month and lasting approximately one month, but may persist for longer at low levels. In acute infection, IgM usually becomes detectable shortly after the rash onset, peaks approximately 7 days after rash onset and remains detectable for 4–12 weeks.3,4
Because rubella incidence in Australia is low, a high proportion of IgM-positive tests will likely be false positive. False-positive serum rubella IgM tests may occur due to the presence of rheumatoid factors (indicating rheumatologic disease) or cross-reacting IgM, or infection with other viruses.
Testing repeat samples in parallel and detection of wild-type rubella virus by PCR can be used to resolve uncertainties in the serologic evaluation of suspected cases. The best specimens for PCR detection are throat or nasopharyngeal swab or urine. Blood can be used but has not been validated.
Particular care should be taken when rubella IgM is detected in a pregnant woman with no history of illness or contact with a rubella-like illness. Pregnant women are often tested for rubella non-immunity as part of their antenatal care. If rubella test results are IgM-positive for persons who have no or low risk of exposure to rubella, additional laboratory evaluation should be considered. Laboratory evaluation is similar to that described in the IgM-positive section of Appendix 1 - Serologic evaluation of pregnant women exposed to rubella.
Objectives of Surveillance
- To monitor the epidemiology of rubella and congenital rubella syndrome in Queensland to inform public health initiatives including vaccination programs.
- To identify cases and outbreaks of rubella to enable appropriate public health responses.
Public Health Significance and Occurrence
Because rubella is usually a mild disease with non-specific symptoms it is often difficult to diagnose clinically. However, rubella is of public health significance because infection during pregnancy can cause miscarriage (up to 20% of cases in the first 8 weeks of gestation), fetal death and congenital anomalies.5
Congenital rubella syndrome occurs in up to 90% of infants born to women who acquire rubella during the first trimester of pregnancy.6 The risk of a single congenital defect falls to approximately 10–20% by the 16th week and defects are rare when the maternal infection occurs after the 20th week of gestation.6 The most common congenital defects are cataracts, heart defects and hearing impairment.7
The incidence of congenital rubella infection has decreased markedly since the schoolgirl vaccination program was initiated in Australia in the early 1970s. Australia achieved elimination status for rubella in 2018.8
Clinical Features
Rubella
Rubella is a mild febrile viral disease with a diffuse, punctate and maculopapular rash. Clinically, this is usually indistinguishable from a febrile rash illness due to measles, dengue, parvovirus B19, human herpesvirus 6, coxsackie virus, echovirus, adenovirus or scarlet fever. The rash occurs in 50%–80% of rubella infected persons.
Children usually present with few or no constitutional symptoms, but adults may experience a 1–5 day prodrome of low grade fever, headache, malaise, mild coryza, and conjunctivitis. Post-auricular, occipital and posterior cervical lymphadenopathy is the most characteristic clinical feature and precedes the rash by 5–10 days. Many cases (up to 50%) are subclinical.
Complications of rubella are not common but occur more often in adults than in children.
Arthralgia or arthritis may occur in up to 70% of adult women who contract rubella but is rare in children and adult males. Fingers, wrists and knees are often affected. Joint symptoms tend to occur about the same time or shortly after appearance of the rash and may last for up to one month; chronic arthritis is rare.
Encephalitis occurs in one in 6,000 cases, more frequently in adults (especially females) than in children. Mortality estimates vary from zero to 50%.
Haemorrhagic manifestations occur in approximately one per 3,000 cases, occurring more often in children than in adults. These may be secondary to low platelets and vascular damage, with thrombocytopenic purpura being the most common. Gastrointestinal, cerebral, or intracranial haemorrhage may occur. Effects may last from days to months, and most patients recover.
Additional complications include orchitis, neuritis and a rare late syndrome of progressive panencephalitis.
Congenital Rubella Syndrome
Rubella is important because it can produce a constellation of anomalies in the developing fetus. Congenital rubella syndrome (CRS) is an illness resulting from embryonic/fetal rubella virus infection.
CRS occurs in up to 90% of infants born to women who are infected with rubella in the first 10 weeks of pregnancy. Infection early in pregnancy has the greatest risk of intrauterine death, spontaneous abortion, and congenital malformations of major organ systems. The risk of defects declines when maternal infection occurs after the 12th week of pregnancy and defects are rare when maternal infection occurs after the 20th week.
The specific signs and symptoms of congenital rubella syndrome may be classified as temporal (e.g. low birth weight); permanent (e.g. deafness); and developmental (e.g. myopia). Common manifestations include cataracts, microphthalmia, congenital glaucoma, microcephaly, meningoencephalitis, developmental delay, patent ductus arteriosus, atrial or ventricular septal defects, purpura, hepatosplenomegaly, jaundice, and radiolucent bone disease. Moderate and severe CRS is usually recognisable at birth. Mild CRS with only slight cardiac involvement or hearing impairment may not be detected for months or even years after birth. Insulin dependent diabetes mellitus in late childhood is a recognised late manifestation of CRS.9
Reservoirs
Humans are the only known reservoir.
Mode of Transmission
Rubella is transmitted via droplet spread and direct contact with nasopharyngeal secretions of people infected with rubella virus.
Vertical transmission can occur when a woman is infected during pregnancy.
Vaccine-associated rubella is not generally transmitted from vaccinated persons to susceptible contacts. The vaccine virus is secreted in breast milk and rare cases of transmission of vaccine virus through breast milk have been reported. However, symptoms in the newborn have been mild or absent and post-partum vaccination of women need not be delayed because of breastfeeding.
Incubation Period
Usual incubation period is 14–17 days (range of 12–23 days).3,6
Period of Communicability
Rubella is highly communicable. The period of communicability is approximately one week before and at least 4 days after the onset of rash. People with rubella are most infectious when the rash is erupting.
Infants with congenital rubella syndrome shed virus in their pharyngeal secretions and urine for up to one year after birth and can transmit the virus to susceptible people caring for them.9
Susceptibility
Immunity is usually permanent after natural infection. 95% of people seroconvert after one dose of rubella-containing vaccine and immunity may be lifelong after vaccination.6 However, rubella reinfection can occur in individuals who have both natural and vaccine-induced immunity.
Re-infection is not associated with a lack of neutralising antibodies, and viraemia is rare. In some studies, reinfection leads only to an IgG booster response, without IgM, whereas in others the appearance of IgM suggests significant viral replication. Clinical rubella has been documented during reinfection of vaccinated and naturally immune individuals. Occasional cases of congenital rubella syndrome after reinfection in pregnancy have been reported. The risk of fetal transmission is probably less than 5% in the first trimester.10
Infants born to immune mothers are ordinarily protected for 6–9 months, depending on the quantity of maternal antibodies.9
Management
Cases
Investigation
In consultation with the attending medical practitioner, ensure laboratory confirmation and all relevant clinical and demographic information are obtained. Ascertain rubella vaccination status.
Laboratory confirmation is recommended for all sporadic cases of rubella and all suspected cases of congenital rubella syndrome.
Restriction
Exclude rubella cases from childcare/school/institution/work until 4 days after onset of rash or until fully recovered, whichever is longer. Avoid contact with non-immune women of childbearing age and pregnant women.
In hospitals, as per current Queensland Health infection control guidelines, patients with suspected or proven rubella should be managed with contact and droplet precautions until 7 days after onset of rash or until fully recovered, whichever is longer. Contact precautions should be applied to infants less than 12 months of age with CRS unless urine and pharyngeal cultures are negative for rubella virus.11
Infants with congenital rubella syndrome should be excluded from childcare and avoid contact with pregnant women for the first 12 months of life unless urine and pharyngeal specimens are negative for rubella virus either by culture or nucleic acid testing after 3 months of age.
Counselling
The case should be advised of the nature of the infection and its mode of transmission. If other vaccinations are incomplete, recommend catch up once recovered.
Pregnant cases
If the patient is pregnant, obtain information regarding number of weeks of gestation at onset of illness. Refer the case promptly to their obstetrician/clinician for counselling regarding the risks of intrauterine rubella infection and appropriate follow-up. If possible, follow up with the referring clinician for pregnancy outcome.
Contacts
Contact tracing
Yes
Definition
Anyone who is likely to have been exposed to the nose or throat secretions of a person with rubella during their infectious period. This may be by direct exposure or droplet spread. Although rubella transmission is usually associated with repeated exposure, transmission has been documented after a single exposure.
Investigation
Prioritise investigation based on the probability of transmission. First priority should be people who share a household with the case, people who attend the same class at school or childcare as the case and people who are work or social contacts of the case. Particular effort should be made to identify all pregnant women who might have been exposed. Pregnant women should ensure they are reviewed by their clinician and serologically tested for rubella.
Except for pregnant women and women who are planning a pregnancy, adequate proof of immunity to rubella includes:
- written documentation of receipt of two doses of a rubella-containing vaccine administered on or after the first birthday
- laboratory evidence of immunity
- birth before 1966.
Women most likely to be sero-negative for rubella include6:
- women born overseas (especially in Asia, Pacific islands, sub-Saharan Africa and South America) who entered Australia after the age of routine vaccination
- Aboriginal and Torres Strait Islander women living in rural and remote regions
- non–English speaking women
- women ≥35 years of age
- Australian-born Muslim women
Pregnant women and women who are planning a pregnancy should be serologically tested.
Any contact who does not have adequate proof of immunity is a susceptible contact.
Management
Non-pregnant contacts
All susceptible non-pregnant contacts aged 12 months and older should be offered a single dose of measles, mumps and rubella (MMR) vaccine, provided there are no contraindications to the vaccine. This will not prevent disease if the contact is already infected. Women who are planning a pregnancy should be advised not to become pregnant for 28 days after receiving the vaccine.
Recommend that all susceptible people avoid further contact with the case for at least 4 days after the onset of rash.
Pregnant contact
Refer pregnant contacts to their clinicians for serologic evaluation of rubella-specific IgM and IgG antibodies (see Appendix 1) and counselling regarding the risks for intrauterine rubella infection. If the woman has an IgG antibody titre below the protective level, or is IgG negative, and remains asymptomatic, a second blood specimen should be collected 28 days after the exposure and tested in parallel with the first to detect subclinical infection. If the woman develops symptoms, specimens for rubella PCR and blood for rubella and other viral serology should be collected and tested as soon as possible to confirm diagnosis of rubella and exclude other relevant viral illnesses.
Susceptible pregnant contacts should avoid further contact with the case, and with close contacts of the case for 6 weeks (approx. 2 incubation periods) after the onset of symptoms of rubella in the case.
Surveillance for rubella and CRS should be enhanced (for example by alerting local clinicians) when confirmed or probable rubella cases are documented in a setting where pregnant women might have been exposed.
Post-exposure prophylaxis for susceptible pregnant contacts with immunoglobulin
Post-exposure prophylaxis (PEP) with NHIG might reduce – but will not eliminate – the risk of rubella. Experimental studies in the 1960s confirmed the efficacy of passive antibody in preventing both viremia and clinical rubella, but there were numerous failures of gamma-globulin to prevent congenital fetal abnormality in actual practice.
Susceptible pregnant contacts of confirmed rubella cases should discuss PEP with their obstetrician/clinician. The effectiveness of PEP with NHIG is most likely within 5 days of rubella exposure.12,13
Serological follow up of recipients is essential. IgM should be done once every 2 weeks for 2 months after last contact with the case to establish whether delayed or subclinical infection occurs.
Administration of NHIG
NHIG is available from Australian Red Cross LifeBlood. The public health unit is responsible for notifying the obstetrician/treating clinician on the process for accessing NHIG for their patient if required. (See the following webpage for order form and contact details for CSL: http://www.blood.gov.au/NHIg (current as at 23 December 2022).
NHIG should be given by deep IM injection; 0.5mL for pregnant contacts up to 160kg and 1mL for those >160kg. The NHIG should be introduced slowly into the muscle, to reduce pain. This product must not be administered intravenously because of possible severe adverse events, and hence an attempt to draw back on the syringe after IM insertion of the needle should be made in order to ensure that the needle is not in a small vessel.6
Contacts in health care settings
Any exposed health care worker who does not have adequate evidence of immunity should be excluded from duty beginning 7 days after exposure to rubella and continuing until either:
- 23 days after last exposure
OR
- 7 days after the onset of rash if illness develops or until fully recovered, whichever is longer.
Susceptible non-pregnant health care workers should be offered MMR vaccine, although post-exposure vaccination will not prevent rubella infection following exposure.
Management of Outbreaks
- Identify potentially susceptible groups and high-risk settings to promote awareness and vaccination.
- Alert GPs, Emergency Departments and childcare facilities to enhance surveillance and case ascertainment.
- Exclude non-immune pregnant women from workplaces with identified transmission.
- Alert hospital infection control practitioners so they can implement the recommendations in health care settings e.g. antenatal clinics.
Preventive Measures
Rubella containing vaccine as MMR is recommended for all infants at 12 months of age, and MMRV (as the second dose of rubella containing vaccine) is recommended for all infants at 18 months of age. Women of childbearing age should be tested prior to pregnancy, if possible. All seronegative women, if not pregnant or intending to become pregnant within 28 days, should be offered MMR vaccine. Opportunities for screening include general practice, antenatal care, family planning services, and fertility and occupational health clinics. All seronegative pregnant women should be offered MMR vaccine post-partum. Opportunities include the postnatal ward and the 6-week postnatal visit. Women should be advised not to become pregnant for 28 days after vaccination.
Anyone born on or after 01/01/1966 who does not have evidence of 2 documented doses of MMR or is seronegative on screening is recommended to receive MMR vaccine.
Use epidemiologic and published data to identify groups with low vaccination coverage (e.g. some migrant groups) and enhance opportunities for vaccination.7
References
- Communicable Disease Network Australia. Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care; 2013 [cited 2023 Aug 23]. Rubella – Surveillance case definition. Available from: https://www.health.gov.au/resources/publications/rubella-surveillance-case-definition?language=en
- Communicable Disease Network Australia. Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care; 2004 [cited 2023 Aug 23]. Rubella (congenital) – Surveillance case definition. Available from: https://www.health.gov.au/resources/publications/rubella-congenital-surveillance-case-definition?language=en
- The Public Health Laboratory Network (PHLN). Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care; 2 [cited 2023 Oct 11]. Rubella – Laboratory case definition. Available from: https://www.health.gov.au/resources/publications/rubella-laboratory-case-definition?language=en
- Tatiana Lanzieri, MD; Susan Redd; Emily Abernathy, MS; Joseph Icenogle, PhD. Rubella - Vaccine Preventable Diseases Surveillance Manual | CDC [Internet]. 2020 [cited 2023 Aug 23]. Available from: https://www.cdc.gov/vaccines/pubs/surv-manual/chpt14-rubella.html
- Bouthry E, Picone O, Hamdi G, Grangeot-Keros L, Ayoubi JM, Vauloup-Fellous C. Rubella and pregnancy: diagnosis, management and outcomes. Prenat Diagn. 2014 Dec;34(13):1246–53.
- Australian Technical Advisory Group on Immunisation (ATAGI). The Australian Immunisation Handbook. 2023 [cited 2023 Aug 23]. The Australian Immunisation Handbook. Available from: https://immunisationhandbook.health.gov.au/home
- Khandaker G, Zurynski Y, Jones C. Surveillance for congenital rubella in Australia since 1993: cases reported between 2004 and 2013. Vaccine. 2014 Nov 28;32(50):6746–51.
- Department of Health and Aged Care. Australian Government Department of Health and Aged Care. Australian Government Department of Health and Aged Care; 2018 [cited 2023 Aug 23]. Rubella officially eliminated from Australia. Available from: https://www.health.gov.au/ministers/the-hon-greg-hunt-mp/media/rubella-officially-eliminated-from-australia
- Reef S, Heymann D. Rubella. In: Heymann D, editor. Control of Communicable Diseases Manual [Internet]. 21 st. American Public Health Association; 2022 [cited 2023 Aug 23]. (Control of Communicable Diseases Manual). Available from: https://ccdm.aphapublications.org/doi/10.2105/CCDM.2745.126
- Coulter C, Wood R, Robson J. Rubella infection in pregnancy [Internet]. Commonwealth Department of Health and Aged Care; 1999 [cited 2023 Sep 29]. Available from: https://www1.health.gov.au/internet/main/publishing.nsf/Content/cda-pubs-cdi-1999-cdi2304-cdi2304a.htm
- National Health and Medical Research Council. Australian Guidelines for the Prevention and Control of Infection in Healthcare [Internet]. 2019. Available from: https://www.nhmrc.gov.au/about-us/publications/australian-guidelines-prevention-and-control-infection-healthcare-2019
- Young MK, Ng SK, Nimmo GR, Cripps AW. The optimal dose of disease-specific antibodies for post-exposure prophylaxis of measles and rubella in Australia: new guidelines recommended. Expert Opin Drug Metab Toxicol. 2018 Jul 3;14(7):663–9.
- Young MK, Cripps AW, Nimmo GR, van Driel ML. Post-exposure passive immunisation for preventing rubella and congenital rubella syndrome. Cochrane Database Syst Rev. 2015 Sep 9;2015(9):CD010586.
Additional resources
Bennett, J, Dolin, R and Blaser, M 2015, Mandell, Douglas and Bennett's Principles and Practice of Infectious Diseases, Elsevier Saunders, Philadelphia.
Begg K, Roche, P, Owen, R, Liu, C et al 2016, ‘Australia's notifiable diseases status, 2016: annual report of the national notifiable diseases surveillance system’, Communicable Disease
Gao, Z, Wood, J, Burgess, M et al 2013, ‘Models of strategies for control of rubella and congenital rubella syndrome - a 40 yr experience from Australia’, Vaccine, vol. 31, no. 4, p. 691-697, doi: 10.1016/j.vaccine.2012.11.043
Ohkusa Y, Sugawara, T Arai, S et al 2014, ‘Short term prediction of the Incidence of Congenital Rubella Syndrome’, Plos Currents, vol. 30, no. 6, retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4234431/
Palasanthiran, P, Starr,M, Jones, C and Giles,M 2022. Management of Perinatal Infections accessed May 2023, available https://asid.net.au/publications
Appendix 1. Serologic evaluation of pregnant women exposed to rubella
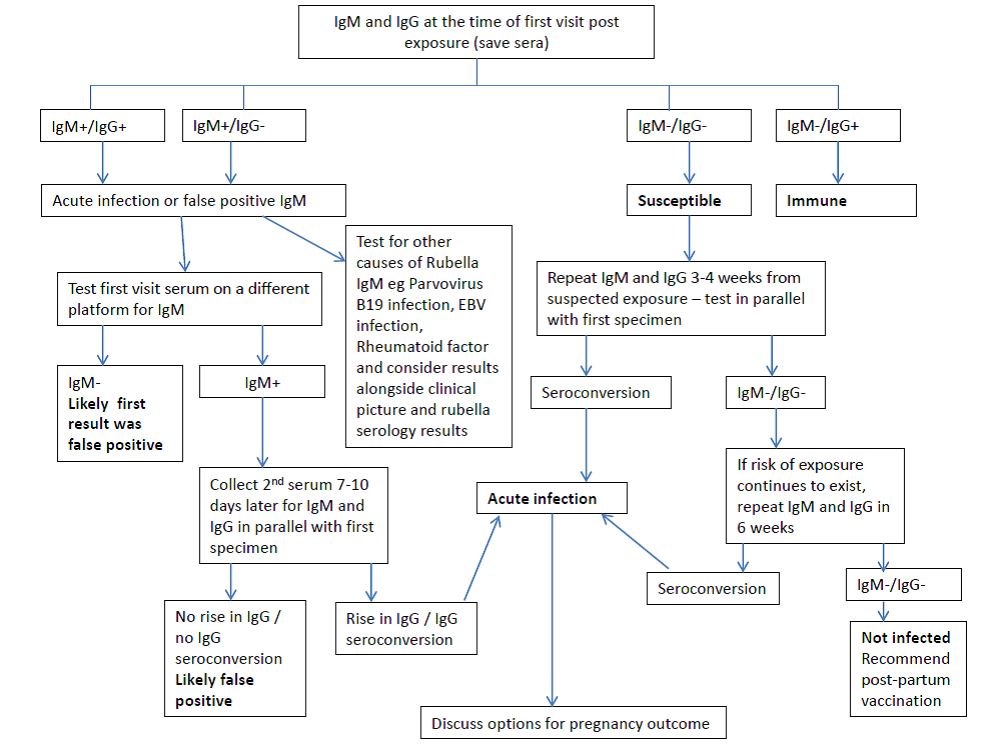
Palasanthiran, P, Starr, M, Jones, C and Giles, M, 2022. Management of Perinatal Infections accessed May 2023, https://asid.net.au/publications
*To note: If a pregnant woman receives NHIG, there is likely to be a transitory rise in rubella IgG. Assessment of serology results after receipt of NHIG should take this into consideration. After receipt of NHIG, it is recommended rubella IgM be measured, by a reference laboratory, once every two weeks for two months after last contact with the case to establish whether delayed or subclinical infection occurs.
Revision History
| Version | Date | Changes |
|---|---|---|
| 1.0 | November 2010 | Full revision of guideline |
| 2.0 | June 2015 | Full revision of guideline |
| 3.0 | June 2019 | Case definitions revision |
| 3.1 | February 2023 | Minor revision and update to case definitions and NHIG recommendations. |

