Cervical screening
From 1 July 2022, changes were made to eligibility for self-collected human papillomavirus (HPV) testing. This follows a recent review of the National Cervical Screening Program which had previously introduced new protocols. Those changes included incorporation of HPV testing and Liquid Based Cytology (LBC). This testing has replaced the Pap smear test which is no longer required.
Screening now tests for the presence of oncogenic HPV. If HPV is detected, reflex LBC will be performed on clinician-collected samples to determine if any cervical cell abnormalities are present. LBC is not able to be performed on self-collected HPV samples.
Self-collection
A patient can be offered the option of self-collection if they are 25 years of age or older, require HPV testing, and have a cervix. Patients who are eligible for self-collection can be provided the following instructions and guidelines:
Self-collected samples are not able to be used for any additional testing such as Chlamydia, Gonorrhoea or Trichomonas PCR.
Patients are not eligible for self-collection if they:
- are under 25 years of age
- were exposed to diethyl-stilbestrol (DES) in utero
- have a history of histologically confirmed AIS
- are symptomatic e.g. is experiencing pain, unusual bleeding, dyspareunia, or unexplained persistent unusual vaginal discharge (especially if malodorous and bloodstained)
- require Test of Cure e.g. following histologically confirmed high-grade squamous intraepithelial lesion (HSIL/CIN II/CIN III) until 2 negative HPV and LBC tests are achieved 12 months apart
- have had a hysterectomy with a history of HSIL.
Self-collection devices
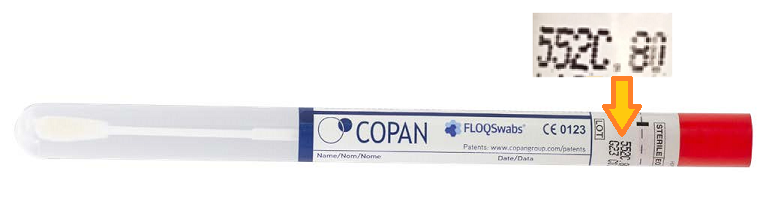
Swabs that have been validated for this test are the Copan 552C.80 and Copan 552C. To determine the type, refer to the following image and the yellow arrow which indicate the location of the swab type and expiry date codes.
Important
Do not cover the swab type code or expiry date with any labels.
Copan 552C.80 Floqswab is our preferred self-collect specimen collection device as we are able to test these in Queensland. This swab MUST be RINSED into a ThinPrep (PreservCyt) vial after collection. Label both the swab (do not cover the type code and expiry date) and ThinPrep vial and send to the laboratory together in the same specimen bag.
Copan 552C Floqswab is still accepted and will be sent interstate for testing. It may take longer to receive results.
S/4HANA reorder codes
- Copan 552C.80 Floqswab: 10439774
- Copan 552C Floqswab: 10031995
Clinician-collection
Sampling protocols for the use of brush, spatula and broom collection devices are available from ThinPrep.
Requesting tests
The request form has been updated to capture specific clinical information which is required for Medicare reimbursement and acceptance of the results into the National Cancer Screening Register. The back of the request form also has eligibility criteria for self-collect samples as well as collection and handling requirements. Contact your local pathology laboratory for hard copies.
Download the cervical screening request form (PDF, 3.0 MB)
Using ieMR or other forms
Digital hospital sites can order tests via ieMR. If ordering in ieMR or using any other request form please specify in clinical notes if the sample is self-collect or clinician-collected.
Need more information?
Email the project team at CervicalCancerScreeningNCSR@health.qld.gov.au.
Refer to the following websites:
- National Cervical Screening Program—Department of Health, Australian Government
- Cervical screening—Queensland Health
- Cancer guidelines wiki—Cancer Council Australia
- Reporting and management guidelines—RCPA (The Royal College of Pathologists of Australasia)